Filter out the noise, focus your attention
With the emergence of big data and machine learning, the pharma, biotech, and life science industries are poised to undergo a data-driven revolution that will accelerate drug discovery and development. High quality machine learning algorithms can scan through thousands of scientific articles, clinical trials, and patient records to identify potential drug targets that were previously unknown. For this you need high quality data solutioning services that will span across drug discovery workflow.

Our services
The life science R&D domain is heavily reliant on data to identify new drug targets, validate clinical trials, and track patient outcomes. Researchers require real-time data on patient outcomes, adverse events, and treatment effectiveness. We provide data solutioning services that support life science researchers in chemistry, biology, and clinical domains.
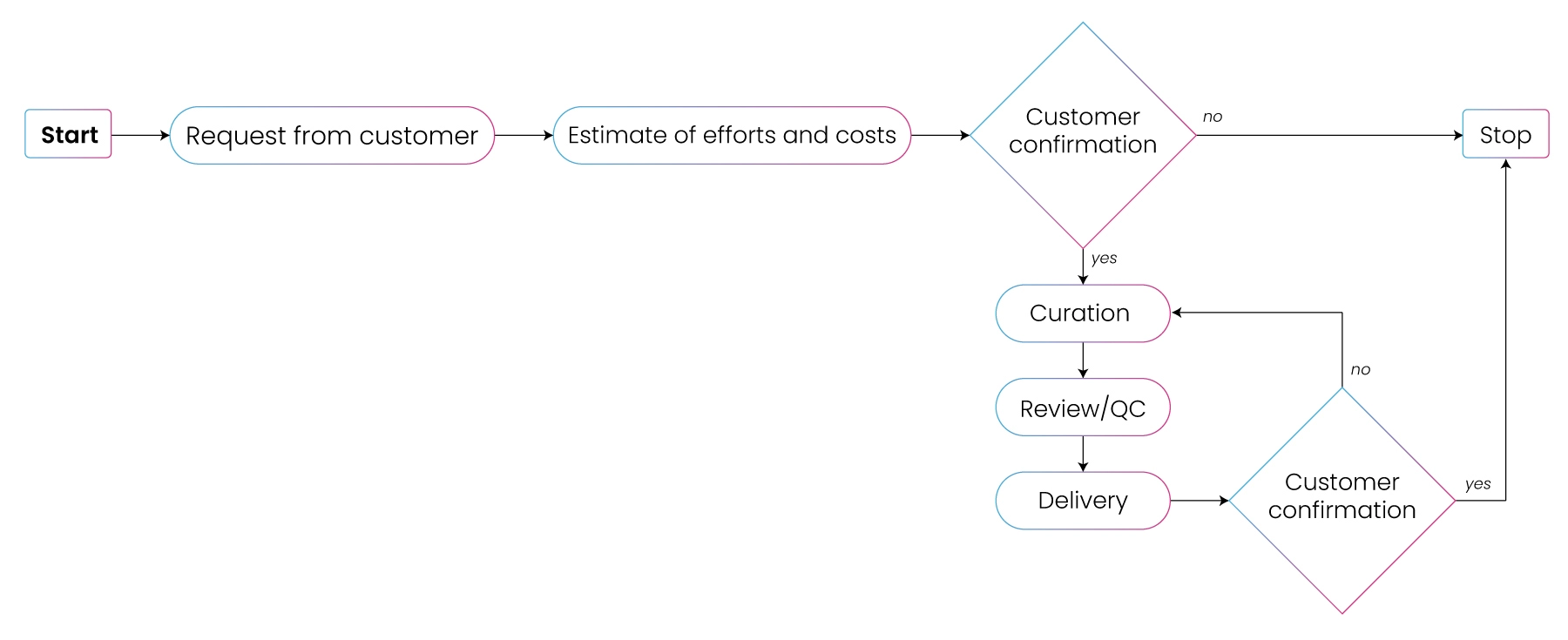
Clinical pharmocology
- Intelligence Quantitative Systems Pharmacology (iQSP)
- Preclinical Toxicology Report Digitization (PTRD)
- National Clinical Trial (NCT) Data Curation
- Legacy Data Standardization Custom Visualizations
Chemistry
- Chemical structure indexing
- Compound-target-indication profiling
- SAR and reaction data curation
- Drug label, safety, and toxicity data exception
- Monoclonal Antibodies →
Healthcare data structuring
- Key opinion leader (KOL) profile data structuring
- Healthcare professional (HCP) landscape mapping for brand marketing
Biology
- Custom dossiers generation for targets and indications
- Actionable mutations for targeted therapy
- Clinical genome variant interpretation and reporting
- Gene and biological process annotation
- Genotype-phenotype mapping
- Multi-omics dataset curation and integration
Case study
Adverse events-related screening
Our client owns a proprietary tool for preclinical toxicity, clinical and pharmacovigilance studies. It required the addition of accurate, validated, up-to-date content on drugs and targets for its adverse events database. We provided data-mining, classification, and excerption services, improving the client’s text-mining pipeline to capture the relevant data points.
Our curation experts manually validated the extracted PMIDs for adverse events. We worked at scale and speed, screening 1000 articles a day. The validated PMIDs were subject to a further round of quality control before being applied to the client’s text mining pipeline to fine-tune its output.
Our services saved the client significant time and resources on the tedious validation process. The client’s database was successfully updated with the validated content twice a week, substantially improving efficiency and text-mining capabilities.
Why Excelra
Holistic Approach
We enable you to go beyond data analytics with our technological expertise helping you to establish an integrated (and secure) data architecture for your drug design and development workflow.
Standardized data set
Our data sets are standardized to meet your requirements, comply with industry standards and regulatory requirements.
Flexible deployment options
Your data, however you want it. On the cloud (AWS, Azure, OpenShift, etc.), on-premises, or data repository.
Knowledge hub
Filter
Ready to get more from data?
Tell us about your objectives. We’ll help get you there.
"*" indicates required fields